Organic reactions {addition reaction} can bind substituents to double bond to make two single bonds.
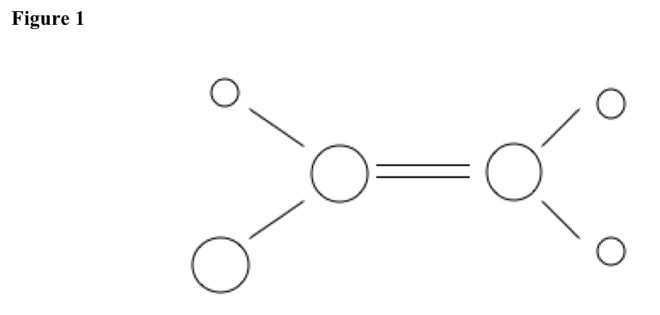
For double bond between two carbons, carbons have two other constituents. Molecule is planar, with constituent angles of 120 degrees. See Figure 1.
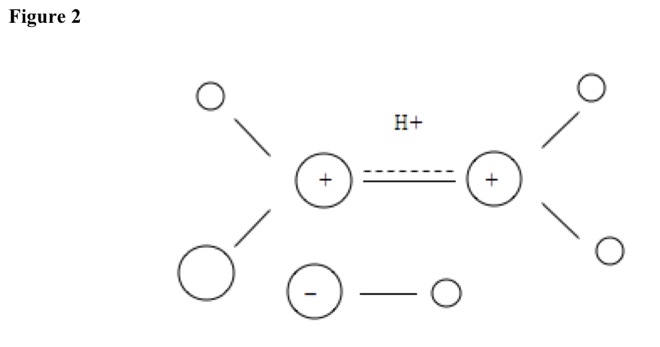
Electrophile, typically hydrogen ion, slowly attacks double-bond electrons to make carbocation. In addition reactions, hydrogen atom binds to primary carbon, by Markownikoff's rule. See Figure 2.
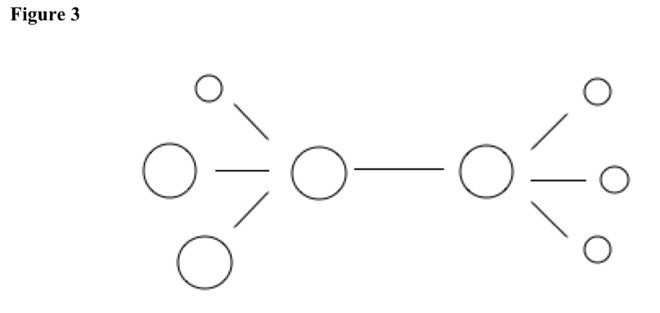
Then nucleophile quickly adds to the other tertiary or secondary carbon, which is more polar than primary carbon atoms. See Figure 3.
conformation
Conformation is typically cis, but is trans for two similar substituents.
polarity
Terminal carbon double bonds are not sufficiently polar for attack, but internal double bonds are polar enough for attack.
alkenes
Alkenes have double bonds between carbons. Alkene + water -> alcohol. Alkene + ammonia -> amine. Alkene + halogen acid -> alkyl halide. Alkene + hydrogen gas -> single bond. Alkene + halogen gas -> alkyl halide, on both carbons with trans conformation.



Physical Sciences>Chemistry>Organic>Chemical Reaction
5-Chemistry-Organic-Chemical Reaction
Outline of Knowledge Database Home Page
Description of Outline of Knowledge Database
Date Modified: 2022.0224